Reproductive Phenology and Breeding Ecology of Panha’s Crocodile Newt, Tylototriton panhai, at Phu Soi Dao National Park, Thailand
DOI:
https://doi.org/10.58837/tnh.26.1.267174Keywords:
Salamandridae, crocodile newt, breeding season, environmental conditions, mark-recaptureAbstract
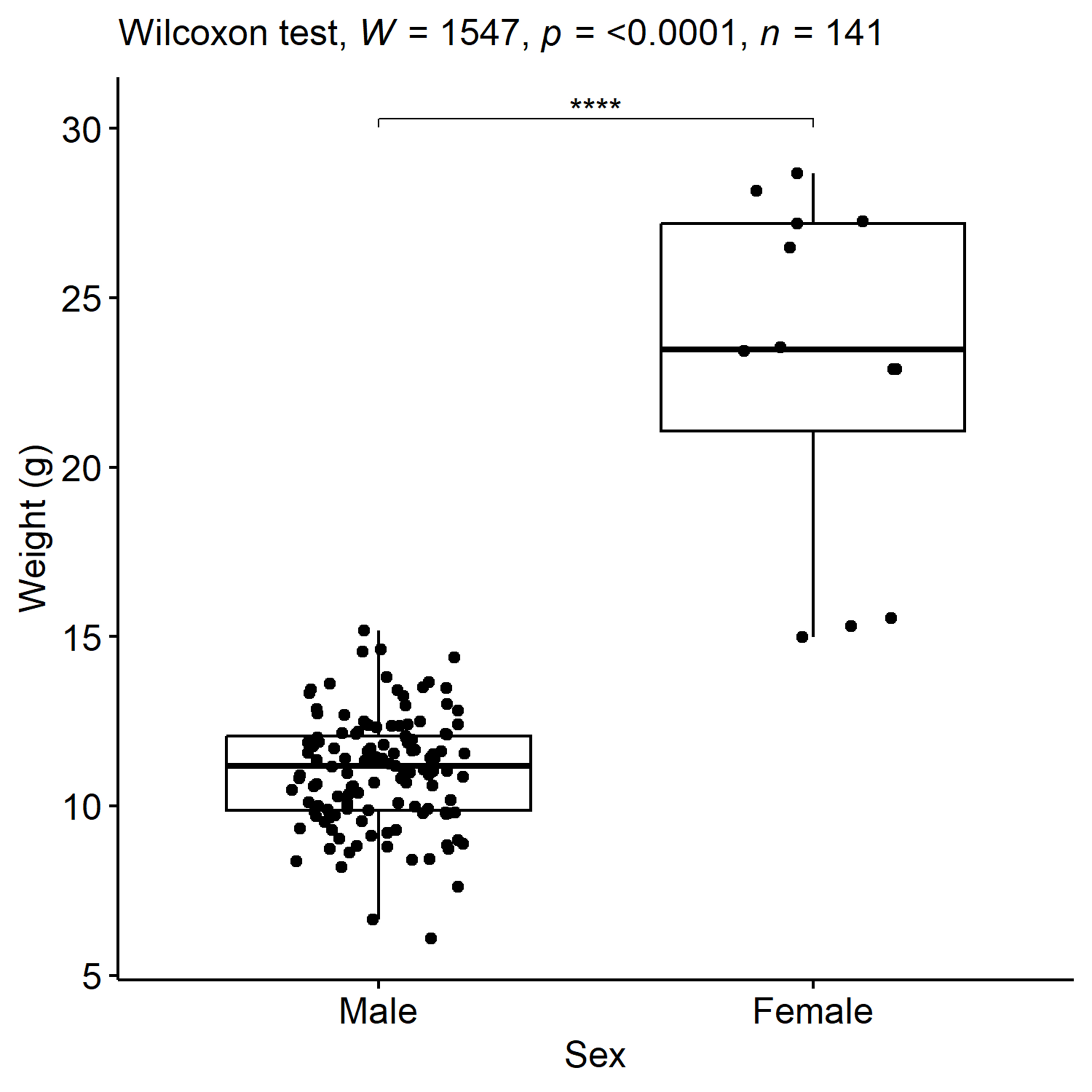
The Panha’s crocodile newt, Tylototriton panhai, has a narrow distribution range in Northeastern Thailand and adjacent Laos PDR. This cryptic species is listed as vulnerable with a decreasing population trend. A local population of T. panhai at Phu Soi Dao National Park, Thailand with a limited distribution through the high mountainous protected area was examined for its reproductive phenology and environmental conditions inducing reproductive period. A total of 269 adult newts (257 males, 12 females) and 298 larvae were captured using a visual encounter survey method, conducted twice a day at daytime and nighttime, and four sets of three-fence arrays with pit fall traps installed at breeding sites and checked daily. The study area (breeding sites) was approximately one square kilometer at 1,633 meters above mean sea level, and was investigated from July 2020 to May 2022. Adult newts were found to be seasonal breeders, with a strongly male biased sex ratio of caught individuals, and visited breeding sites only in the wet season (May–October) with the exception of one male recorded in November 2021. Males stayed at breeding sites for several months (May–November), whereas females visited for a shorter period of time (May to July). Numbers of adults decreased in the breeding sites after breeding, whereas larvae were initially observed in June and had been found in seasonal aquatic habitats until early dry season. Both adults and larvae disappeared in March 2021. A quasi-Poisson regression model showed a significant correlation between the number of adult newts visiting breeding sites and air temperature, humidity, or water pH. Furthermore, drought-induced water scarcity influenced the duration that adults and larvae remained in aquatic habitats. Adult newts were weighed and measured for 16 morphometric characters. Females were larger in body size and exhibited larger traits than males, as shown by ANCOVA with snout–vent length as a covariate (p < 0.05). A total of 108 adults (99 males, 9 females) were tagged with a 7-mm PIT tag, from which 20 recaptured males (20.20%) were found from May, 2021–April, 2022. The longest duration of the same male in aquatic habitats was from May to November in 2021, but no females were recaptured in the aquatic habitats, suggesting either lower site fidelity, different habitat use, or lower survival of females during this period. The estimated population size using RMark with the POPAN model was 249 individuals with an increasing abundance trend (227–249), and the estimate detection probability for males (0.26 ± 0.04) was higher than for females (0.02 ± 0.01).
References
AmphibiaWeb. 2024. AmphibiaWeb: Information on amphibian biology and conservation. University of California, Berkeley. Available from: https://amphibiaweb.org, (September 20, 2025).
Anderson, J. 1871. Description of a new genus of newts from western Yunan. In Proceedings of the Zoological Society of London, 1871: 423–425.
Anderson, T.L., Ousterhout, B.H., Peterman, W.E., Drake, D.L. and Semlitsch, R.D. 2015. Life history differences influence the impacts of drought on two pond‐breeding salamanders. Ecological Applications, 25(7): 1896–1910.
Bagnouls, F. and Gaussen, H. 1957. Les climats biologiques et leur classification. Annales de Géographie, 66: 193–220.
Bedi, A., Bedi, V., Nag, S. and Suyesh, R. 2021. Wild observations of the reproductive behaviour and first evidence of vocalization in Crocodile newt Tylototriton himalayanus (Caudata: Salamandridae) from the Himalayan biodiversity hotspot in Eastern India. Salamandra, 57(1): 65–74.
Bernardes, M., Rödder, D., Nguyen, T.T., Pham, C.T., Nguyen, T.Q. and Ziegler, T. 2013. Habitat characterization and potential distribution of Tylototriton vietnamensis in northern Vietnam. Journal of Natural History, 47(17–18): 1161–1175.
Bernardes, M., Rauhaus, A., Michel, C., Nguyen, T.Q., Le, M.D., Pasmans, F. and Ziegler, T. 2017. Larval development and breeding ecology of Ziegler's Crocodile Newt, Tylototriton ziegleri Nishikawa, Matsui and Nguyen, 2013 (Caudata: Salamandridae), compared to other Tylototriton representatives. Amphibian and Reptile Conservation, 11(1): 72–87.
Bӧhme, W., Schottler, T., Nguyen, T.Q. and Kohler, J. 2005. A new species of salamander, genus Tylototriton (Urodela: Salamandridae), from northern Vietnam. Salamandra, 41(4): 215–220.
Bull, J.J. and Shine, R. 1979. Iteroparous animals that skip opportunities for reproduction. The American Naturalist, 114(2): 296–303.
Dervo, B.K., Baerum, K.M., Skurdal, J. and Museth, J. 2016. Effects of temperature and precipitation on breeding migrations of amphibian species in southeastern Norway. Scientifica, 2016: 1–8.
Dodd, C.K. (Ed.). 2010. Amphibian ecology and conservation: a handbook of techniques. Oxford University Press Inc., New York, 584 pp.
Dufresnes, C., and Hernandez, A. 2022. Towards completing the crocodile newts’ puzzle with all-inclusive phylogeographic resources. Zoological Journal of the Linnean Society 197: 620–640.
Ficetola, G.F. and Maiorano, L. 2016. Contrasting effects of temperature and precipitation change on amphibian phenology, abundance and performance. Oecologia, 181: 683–693.
Frost, D. R. 2022. Amphibian Species of the World: an Online Reference. Version 6.2, American Museum of Natural History, New York, USA. Available from: https://amphibiansoftheworld.amnh.org/index.php, (March 1, 2022). doi.org/10.5531/db.vz.0001
Fu, V.W.K., Karraker, N.E. and Dudgeon, D. 2013. Breeding dynamics, diet, and body condition of the Hong Kong newt (Paramesotriton hongkongensis). Herpetological Monographs, 27(1): 1–22.
Geiger, R. 1961. Überarbeitete Neuausgabe von Geiger, R.: Köppen-Geiger / Klima der Erde. (Wandkarte 1:16 Mill.), Darmstadt.
Glime, J.M. and Boelema, W.J. 2017. Amphibians: Anuran adaptations. Chapt. 14-1. In: Glime, J.M. (Ed.). Bryophyte Ecology. Bryophyte ecology. Volume 2. Bryological Interaction. Michigan Technological University, Houghton, 1–29.
Grayson, K.L., Bailey, L.L. and Wilbur, H.M. 2011. Life history benefits of residency in a partially migrating pond‐breeding amphibian. Ecology, 92(6): 1236–1246.
Grayson, K.L., De Lisle, S.P., Jackson, J.E., Black, S.J. and Crespi, E.J. 2012. Behavioral and physiological female responses to male sex ratio bias in a pond-breeding amphibian. Frontiers in Zoology, 9(1): 1–10.
Griffiths, R.A. 1986. Feeding niche overlap and food selection in smooth and palmate newts, Triturus vulgaris and T. helveticus, at a pond in mid-Wales. The Journal of Animal Ecology, 55(1): 201–214.
Halliday, T.R. and Verrell, P.A. 1986. Sexual selection and body size in amphibians. Herpetological Journal, 1(3): 86–92.
Halliday, T. R. and Tejedo, M. 1995. Intrasexual selection and alternative mating behaviour. In: Heatwole, H. and Sullivan, B.K. (Eds). Amphibian Biology, Social Behaviour, Surrey Beatty, Chipping Norton, 419–468.
Hedrick, A.V. and Temeles, E.J. 1989. The evolution of sexual dimorphism in animals: hypotheses and tests. Trends in Ecology and Evolution, 4(5): 136–138.
Hernandez, A. 2016. Crocodile Newts: The Primitive Salamandridae of Asia: Genera Echinotriton and Tylototriton. Chimaira Editions, Frankfurt, 415 pp.
Hernandez, A. 2017. New localities for Tylototriton panhai and Tylototriton uyenoi Nishikawa, Khonsue, Pomchote, and Matsui 2013 in northern Thailand. Bulletin de la Société Herpétologique de France, 162: 110–112.
Hernandez, A., Escoriza, D. and Hou, M. 2018. Patterns of niche diversification in south-east Asian crocodile newts. Zoologischer Anzeiger, 276: 86–93.
Hernandez, A., Escoriza, D., Pomchote, P. and Hou, M. 2019. New localities for Tylototriton uyenoi, T. panhai and T. anguliceps in Thailand with remarks on the southernmost distribution of the genus. Herpetological Bulletin, 147:15–18.
Hernandez, A., and Pomchote, P. 2020. Habitat, distribution and life history of the polytypic Panha’s Crocodile Newt, Tylototriton panhai in northeastern Thailand. Alytes, 37(3–4): 25–46.
Hernandez, A., Pomchote, P. and Jamin, A. 2022. First reproduction of Panha’s crocodile newt Tylototriton panhai in captivity, with a description of the courtship behaviour, eggs and larval development. Herpetological Bulletin, 159: 6–11.
Heyer, W.R., Donnelly, M.A., McDiarmid, R.W., Hayek, L.C. and Foster, M.S. 1994. Measuring and monitoring biological diversity: standards methods for amphibians, Smithsonian Institution Press, Washington, 384 pp.
IUCN. 2001. IUCN Red List categories and criteria: version 3.1. Prepared by the IUCN Species Survival Commission. IUCN, Gland, Switzerland and Cambridge, United Kingdom, 30 pp.
Johnson, E.R., Bowerman, B.L., Thomas, M.A., Thompson, L.M. and Grayson, K.L. 2017. The influence of environmental factors on pond activity of aquatic red-spotted newts Notophthalmus viridescens. Journal of Freshwater Ecology, 32(1): 711–720.
Kottek, M., Grieser, J., Beck, C., Rudolf, B. and Rubel, F. 2006. World map of the Köppen-Geiger climate classification updated. Meteorologische Zeitschrift, 15(3): 259–263.
Köppen, W. 1900. Versuch einer Klassifikation der Klimate, vorzugsweise nach ihren Beziehungen zur Pflanzenwelt. Geographische Zeitschrift, 6(12): 657–679.
Kuzmin, S.L., Dasgupta, R. and Smirina, E.M. 1994. Ecology of the Himalayan newt (Tylototriton verrucosus) in Darjeeling Himalayas, India. Russian Journal of Herpetology, 1(1): 69–76.
Le, D.T., Nguyen, T.T., Nishikawa, K., Nguyen, S.L.H., Van Pham, A., Matsui, M. and Nguyen, T.Q. 2015. A new species of Tylototriton Anderson, 1871 (Amphibia: Salamandridae) from northern Indochina. Current Herpetology, 34(1): 38–50.
Lyu, Z.T., Wang, J., Zeng, Z.C., Zhou, J.J., Qi, S., Wan, H. and Wang, Y.Y. 2021. A new species of the genus Tylototriton (Caudata, Salamandridae) from Guangdong, southern China, with discussion on the subgenera and species groups within the genus. Vertebrate Zoology, 71: 697–710.
Maiorana, V.C. 1976. Size and environmental predictability for salamanders. Evolution, 30: 599–613.
Marangio, M.S. and Anderson, J.D. 1977. Soil moisture preference and water relations of the marbled salamander, Ambystoma opacum (Amphibia, Urodela, Ambystomatidae). Journal of Herpetology, 11: 169–176.
Nishikawa, K., Khonsue, W., Pomchote, P. and Matsui, M. 2013a. Two new species of Tylototriton from Thailand (Amphibia: Urodela: Salamandridae). Zootaxa, 3737(3): 261–279.
Nishikawa, K., Matsui, M. and Nguyen, T.T. 2013b. A new species of Tylototriton from northern Vietnam (Amphibia: Urodela: Salamandridae). Current Herpetology, 32(1): 34–49.
Packer, W.C. 1960. Bioclimatic influences on the breeding migration of Taricha rivularis. Ecology, 41(3): 509–517.
Phimmachak, S., Aowphol, A. and Stuart, B.L. 2015a. Morphological and molecular variation in Tylototriton (Caudata: Salamandridae) in Laos, with description of a new species. Zootaxa, 4006(2): 285–310.
Phimmachak, S., Stuart, B.L., and Aowphol, A. 2015b. Ecology and natural history of the knobby newt Tylototriton podichthys (Caudata: Salamandridae) in Laos. Raffles Bulletin of Zoology, 63(2015): 389–400.
Pimentel, R.A. 1960. Inter-and intrahabitat movements of the rough-skinned newt, Taricha torosa granulosa (Skilton). American Midland Naturalist, 63(2): 470–496.
Pomchote, P., Khonsue, W., Sapewisut, P., Eto, K. and Nishikawa, K. 2020b. Discovering a population of Tylototriton verrucosus (Caudata: Salamandridae) from Thailand: implications for conservation. Tropical Natural History, 20(1): 1–15.
Pomchote, P., Khonsue, W., Thammachoti, P., Hernandez, A., Peerachidacho, P., Suwannapoom, C., Onishi, Y. and Nishikawa, K. 2020a. A new species of Tylototriton (Urodela: Salamandridae) from Nan Province, northern Thailand. Tropical Natural History, 20(2): 144–161.
Pomchote, P., Peerachidacho, P., Hernandez, A., Sapewisut, P., Khonsue, W., Thammachoti, P. and Nishikawa, K. 2021. A new species of the genus Tylototriton (Urodela, Salamandridae) from western Thailand. ZooKeys, 1072: 83–105.
Pongthornpruek, S., Pampasit, S., Promtep, K. and Nabheerong, P. 2008. The diversity of pteridophytes along a natural trial at Phu Soi Dao National Park, Phitsanulok province, Thailand. Asia-Pacific Journal of Science and Technology, 13(8): 895–905.
Pough, F.H., Andrews, R.M., Cadle, J.E., Crump, M.L., Savitzky, A.H. and Wells, K.D. 1998. Herpetology (Vol. 830). Prentice Hall, Upper Saddle River, New Jersey, 577 pp.
Raffaëlli, J. 2022. Salamanders & Newts of the World. Plumelec, France: Penclen Édition.
R Core Team. 2022. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/.
Schwarz, C.J. and Arnason, A.N. 1996. A general methodology for the analysis of capture-recapture experiments in open populations. Biometrics, 52: 860–873.
Scott, N.J. and Woodward, B.D., 1994. Surveys at breeding sites. Measuring and monitoring biological diversity: standard methods for amphibians. In: Heyer, R., Maureen, A., Donnelly, M., Mercedes, F. and Mcdiarmid, R. (Eds.). Measuring and Monitoring Biological Diversity: Standard methods for amphibians, Smithsonian Institution Press, Washington, 118–125.
Seglie, D., Roy, D. and Giacoma, C. 2010. Sexual dimorphism and age structure in a population of Tylototriton verrucosus (Amphibia: Salamandridae) from the Himalayan Region. Copeia, 2010(4): 600–608.
Semlitsch, R.D. and Wilbur, H.M. 1988. Effects of pond drying time on metamorphosis and survival in the salamander Ambystoma talpoideum. Copeia, 1988: 978–983.
Sherman, E. and Van Munster, K. 2012. Pond pH, acid tolerance, and water preference in newts of Vermont. Northeastern Naturalist, 19(1): 111–122.
Shine, R. 1979. Sexual selection and sexual dimorphism in the Amphibia. Copeia, 1979: 297–306.
Smith, D.C. 1983. Factors controlling tadpole populations of the chorus frog (Pseudacris triseriata) on Isle Royale, Michigan. Ecology, 64(3): 501–510.
Sparreboom, M. 2014. Salamanders of the Old World: the salamanders of Europe, Asia and northern Africa. Royal Dutch Society for Natural History Publishers, Zeist, 431 pp.
Taylor, E. H. 1962. The amphibian fauna of Thailand. The University of Kansas Science Bulletin, 43: 265–599.
Todd, B.D. and Winne, C.T. 2006. Ontogenetic and interspecific variation in timing of movement and responses to climatic factors during migrations by pond-breeding amphibians. Canadian Journal of Zoology, 84(5): 715–722.
Vitt, L.J. and Caldwell, J.P. 2013. Herpetology: an introductory biology of amphibians and reptiles. Academic press, Amsterdam, 720 pp.
Welsh-Appleby, B. 2015. Analysis of persistent marking techniques using passive integrated transponders and visible im-plant elastomer through metamorphosis in Ambystoma mavortium. Environmental Studies Undergraduate Student Thesis, University of Nebraska, Lincoln, 175 pp.
Whiteman, H.H., Doyle, J.M., Aubee, C., Brown, R., Thomason, S. and Schoborg, T. 2016. A PIT tagging technique for ambystomatid salamanders. Herpetological Review, 47: 32–34.
Wells, K.D. 2010. The ecology and behavior of amphibians, University of Chicago press, Chicago (Illinois), 1400 pp.
Wilcox, J.T., Vang, C.D., Muller, B.R. and Alvarez, J.A. 2017. Drought influences reproductive timing in two newt (Taricha) congeners. Herpetology Notes, 10: 585–587.
Downloads
Additional Files
Published
How to Cite
Issue
Section
Categories
License
Chulalongkorn University. All rights reserved. No part of this publication may be reproduced, translated, stored in a retrieval system, or transmitted in any form or by any means, electronic, mechanical, photocopying, recording or otherwise, without prior written permission of the publisher











