Testis-Ova in Male Rice Frog (Fejervarya limnocharis) - A Natural Process of Sex Differentiation or a Suitable Biomarker of Effect for Ecotoxicology?
DOI:
https://doi.org/10.58837/tnh.26.1.267836Keywords:
amphibian, endocrine disrupting chemicals, histology, intersex, rice fieldAbstract
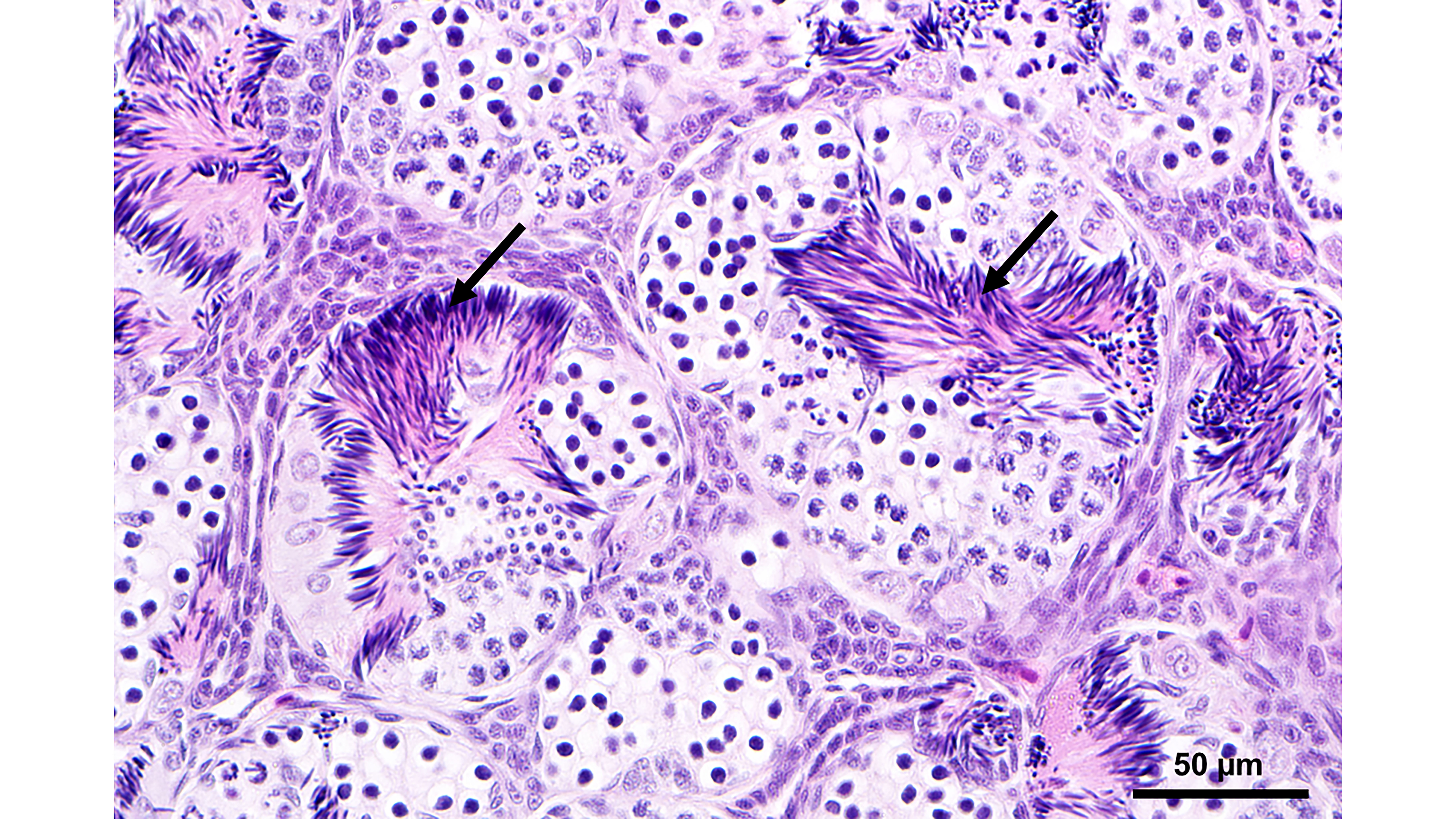
A previous study found that adult male rice frogs (Fejervarya limnocharis) inhabiting agricultural areas with varying levels of herbicide use exhibited testis-ova (TO). However, it remained unclear whether the occurrence of TO is associated with underlying herbicide contamination in agricultural areas or represents a natural aspect of testicular development in this species. This study aimed to examine the gonadal histology and intersex condition of the male rice frog, F. limnocharis, living in an agricultural area in central Thailand with no history of herbicide utilization over the last 10 years. The results showed that the incidences of testicular ovarian follicles (TOFs) in adult, subadult, and juvenile frogs were 46/50 (92%), 7/7 (100%), and 6/6 (100%), respectively. Crucially, these TOFs were observed in rice frogs even when herbicide analysis showed paraquat tissue residue below detection limits. These findings suggest that the presence of oocytes within the testis in F. limnocharis may represent a natural, transition phase of testicular development in the undifferentiated and semi-differentiated types of the normal sex differentiation process in amphibians. This prompted an evaluation of whether the TOFs could serve as a biomarker for assessing the effect of and susceptibility to herbicide contamination in this frog species.
References
Beebee, T.J.C. and Griffiths, R.A. 2005. The amphibian decline crisis: A watershed for conservation biology?. Biological Conservation, 125: 271-285.
Brown, P., Charlton, A., Cuthbert, M., Barnett, L., Ross, L., Green, M., Gillies, L., Shaw, K., and Fletcher, M. 1996. Identification of pesticide poisoning in wildlife. Journal of Chromatography A, 754(1-2):463–478.
Coady, K.K., Murphy, M.B., Villeneuve, D.L., Hecker, M., Jones, P.D., Carr, J.A., Solomon, K.R., Smith, E.E., Van Der Kraak, G., Kendall, R.J., and Giesy, J.P. 2005. Effect of atrazine on metamorphosis, growth, laryngeal and gonadal development, aromatase activity and sex steroid concentrations in Xenopus laevis. Ecotoxicology and Environmental Safety, 62: 160-173.
Eastwood, S. and Couture, P. 2002. Seasonal variations in condition and liver metal concentrations of yellow perch (Perca flavescens) from a metal-contaminated environment. Aquatic Toxicology, 58: 43-56.
Felsemburgh, F.A., Carvalho-e-Silva, S.P. and de Brito-Gitirana, L. 2007. Morphological characterization of the anuran integument of the Proceratophrys and Odontophrynus genera (Amphibia, Anuran, Leptodactylidae). Micron 38, 439-445.
Gramapurohit, N.P., Shanbhag, B.A. and Saidapur, S.K. 2000. Pattern of gonadal sex differentiation, development, and onset of steroidogenesis in the frog, Rana curtipes. General and Comparative Endocrinology, 119: 256-264.
Hayes, T.B., Collins, A., Lee, M., Mendoza, M., Noriega, N., Stuart, A.A. and Vonk, A. 2002. Hermaphroditic, demasculinized frogs after exposure to the herbicide atrazine at low ecologically relevant doses. Proceedings of the National Academy of Science USA 99, 5476-5480.
Jantawongsri, K. 2014. Immune response of rice frog Fejervarya limnocharis (Gravenhorst, 1829) in herbicide utilized agricultural area at Wiang Sa district, Nan province. Master's thesis, Chulalongkorn University.
Jantawongsri, K., Thammachoti, P., Kitana, J., Khonsue, W., Varanusupakul, P. and Kitana, N. 2015. Altered immune response of the rice frog Fejervarya limnocharis living in agricultural area with intensive herbicide utilization at Nan Province, Thailand. EnvironmentAsia, 8: 68-74.
Kitana, J., Butsiri, R., Thammachoti, P., Jantawongsri, K., Khonsue, W. and Kitana, N. 2018. Intersex condition in the male rice frog Fejervarya limnocharis - natural process of gonad development or valid concern for ecotoxicologists?. Abstract, the 11th Society of Environmental Toxicology and Chemistry - Asia Pacific 2018, 82-84.
Kobayashi, T., Kumakura, M., Yoshie, S., Sugishima, T. and Horie, Y. 2015. Dynamics of testis-ova in a wild population of Japanese pond frogs, Rana nigromaculata. Journal of Experimental Zoology Part A: Ecological and Integrative Physiology, 323(2): 74-79. doi: 10.1002/jez.1898
Maneein, R., Khonsue, W., Varanusupakul, P. and Kitana, N. 2011. Association between atrazine utilization and biologic response of rice field crab Esanthelphusa nani in paddy fields of Nan Province, Thailand. Research Journal of Chemistry and Environment, 15: 1018-1023.
Mali, P.V and Gramapurohit, N.P. 2015. Pattern of gonadal differentiation and development up to sexual maturity in the frogs, Microhyla ornata and Hylarana malabarica: A comparative study. Journal of Experimental Zoology Part A: Ecological and Integrative Physiology, 323: 666-678.
Ogielska, M., Chmielewska, M., and Rozenblut-Kościsty, B. 2024. Pregametogenesis: The earliest stages of gonad and germline differentiation in anuran amphibians. Biology, 13(12): 1017.
Othman, M.S. 2009. Using the rice frog (Fejervarya limnocharis) as a sentinel species for cadmium contamination in Tak Province, Thailand. Doctoral dissertation, Chulalongkorn University.
Othman, M.S., Khonsue, W., Kitana, J., Thirakhupt, K., Robson, M.G. and Kitana, N. 2016. Morphometric and gravimetric indices of two populations of rice frog (Fejervarya limnocharis) naturally exposed to different environmental cadmium levels. Journal Sains Kesihatan Malaysia, 14(2): 57-64.
Phuge, S.K. and Gramapurohit, N.P. 2013. Gonadal sex differentiation, development up to sexual maturity and steroidogenesis in the skipper frog, Euphlyctis cyanophlyctis. General and Comparative Endocrinology, 181: 65-71.
Quick, M.P., Dyson, D.A., and Holliman, A. 1990. Acute and sub-acute paraquat poisoning in a pack of foxhounds. Science and Justice, 30(6): 371-376.
Saber, S., Tito, W., Said, R., Mengistou, S. and Alqahtani, A. 2017. Amphibians as bioindicators of the health of some wetlands in Ethiopia. The Egyptian Journal of Hospital Medicine, 66(1): 66-73.
Storrs-Méndez, S.I. and Semlitsch, R.D. 2010. Intersex gonads in frogs: understanding the time course of natural development and role of endocrine disruptors. Journal of Experimental Zoology. Part B, Molecular and developmental evolution, 314(1): 57-66.
Tanimura, A. and Iwasawa, H. 1989. Origin of somatic cells and histogenesis in the primordial gonad of the japanese tree frog Rhacophorus arboreus. Anatomy and Embryology, 180: 165-173.
Thammachoti, P., Khonsue, W., Kitana, J., Varanusupakul, P. and Kitana, N. 2012. Morphometric and gravimetric parameters of the rice frog Fejervarya limnocharis living in areas with different agricultural activity. Journal of Environmental Protection, 2: 1403-1408.
Torreilles, S.L., McClure, D.E. and Green, S.L. 2009. Evaluation and refinement of euthanasia methods for Xenopus laevis. Journal of the American Association for Laboratory Animal Science, 48: 512-516.
Traijitt, T., Kitana, N. and Kitana, J. 2020. Pattern of gonadal sex differentiation in the rice field frog Hoplobatrachus rugulosus (Anura: Dicroglossidae). Zoological Studies, 59:e51.
Witschi, E. 1929. Studies of sex differentiation and sex determination in amphibians. I. Development and sexual differentiation of the gonads of Rana sylvatica. Journal of Experimental Zoology, 52: 235-265.
Downloads
Published
How to Cite
Issue
Section
Categories
License
Chulalongkorn University. All rights reserved. No part of this publication may be reproduced, translated, stored in a retrieval system, or transmitted in any form or by any means, electronic, mechanical, photocopying, recording or otherwise, without prior written permission of the publisher











