Comparative Analysis of Activated Carbon Derived from Coconut Shell and Oil Palm Biomass at Various Carbonization Temperatures
Main Article Content
Abstract
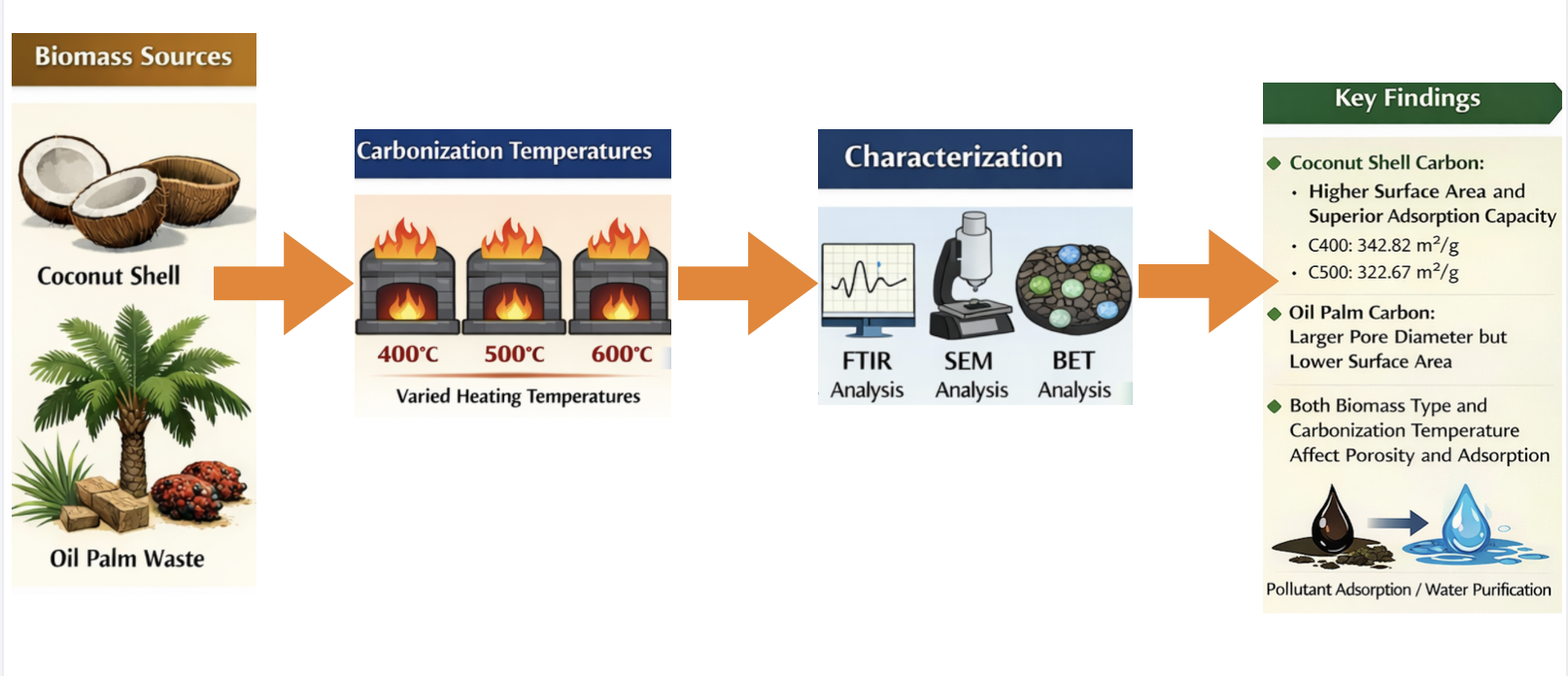
This study compared the optimal conditions for producing activated carbon from coconut shell and oil palm biomass via chemical activation using 70% H2SO4. Carbonization was conducted at 400, 500, and 600 °C for 2 h. At 600 °C, partial combustion occurred in both precursors, leading to ash formation, whereas no such effects were observed at 400 °C and 500 °C. Therefore, activated carbons obtained at 400 °C and 500 °C were further characterized using Fourier Transform Infrared (FTIR) Spectroscopy, Scanning Electron Microscopy (SEM), and Brunauer–Emmett–Teller (BET) surface area analysis. FTIR spectra of both coconut shell and palm-derived samples showed broad O–H stretching bands around 3440–3450 cm-1 and peaks at 1610–1630 cm-1 corresponding to C=O and C=C stretching vibrations. Notably, the palm-based sample carbonized at 500 °C displayed slight variations in its FTIR spectra compared with the other three conditions. Consistently, SEM images revealed that the P500 sample exhibited distinct surface morphologies compared with the others. BET analysis further indicated that the specific surface areas of C400, C500, P400, and P500 were 342.82, 322.67, 196.93, and 208.13 m²/g, respectively, demonstrating that coconut shell–derived carbons developed substantially higher surface areas than palm-based carbons. The high surface areas and well-developed porosity of these activated carbons suggest their suitability for applications in wastewater treatment, heavy metal and organic pollutant adsorption, and potential use as adsorbents in catalysis or separation processes.
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
The content and information in the article published in Journal of Rajamangala University of Technology Srivijaya It is the opinion and responsibility of the author of the article. The editorial journals do not need to agree. Or share any responsibility.
References
Ahmed, M.B., Zhou, J.L., Ngo, H.H. and Guo, W. 2016. Insight into biochar properties and its cost analysis. Bioresource Technology 214: 836-851. DOI: https://doi.org/10.1016/j.biortech.2016.05.057
Ajien, A., Idris, J., Md Sofwan, N., Husen, R. and Seli, H. 2023. Coconut shell and husk biochar: A review of production and activation technology, economic, financial aspect and application. Waste Management & Research 41(1): 37-51. DOI: https://doi.org/10.1177/0734242X221127167
Chew, T.W., H’Ng, P.S., Luqman Chuah Abdullah, B.C.T.G., Chin, K.L., Lee, C.L., Mohd Nor Hafizuddin, B.M.S. and TaungMai, L. 2023. A review of bio-based activated carbon properties produced from different activating chemicals during chemical activation process on biomass and its potential for Malaysia. Materials 16(23): 7365. DOI: https://doi.org/10.3390/ma16237365
Foo, K.Y. and Hameed, B.H. 2012. Preparation, characterization and evaluation of adsorptive properties of orange peel based activated carbon via microwave induced K2CO3 activation. Bioresource Technology 104: 679-686. DOI: https://doi.org/10.1016/j.biortech.2011.10.005
Gratuito, M.K.B., Panyathanmaporn, T., Chumnanklang, R.-A., Sirinuntawittaya, N. and Dutta, A. 2008. Production of activated carbon from coconut shell: Optimization using response surface methodology. Bioresource Technology 99: 4887–4895. DOI: https://doi.org/10.1016/j.biortech.2007.09.042
Herawan, S.G., Hadi, M.S., Ayob, M.R. and Putra, A. 2013. Characterization of activated carbons from oil-palm shell by CO2 activation with no holding carbonization temperature. The Scientific World Journal 624865: 1-6. DOI: https://doi.org/10.1155/2013/624865
Ioannidou, O. and Zabaniotou, A. 2007. Agricultural residues as precursors for activated carbon productionA review. Renewable and Sustainable Energy Reviews 11(9): 1966-2005. DOI: https://doi.org/10.1016/j.rser.2006.03.013
Kundu, S., Khandaker, T., Mia Anik, M.A.-A., Hasan, M.K., Dhar, P.K., Dutta, S.K., Latif, M.A. and Hossain, M.S. 2024. A comprehensive review of enhanced CO2 capture using activated carbon derived from biomass feedstock. RSC Advances 14: 29693. DOI: https://doi.org/10.1039/D4RA04537H
Lee, C.L., Chin, K.L., H’Ng, P.S., Hafizuddin, M.S. and Khoo, P.S. 2023. Activation temperature and particle size of palm kernel shell vs. the surface properties of activated carbon. Bioresources 18(2): 345-358. DOI: https://doi.org/10.15376/biores.18.1.1714-1730
Nilufur, A.B., Thirumalaisamy, R. and Harini, S. 2023. Activated carbon from coconut shell: Synthesis and its commercial application-A recent review. Applied Science and Engineering Progress 16(2): 1652.
Rao, M.M., Ramesh, A., Rao, G.P.C. and Seshaiah, K. 2006. Removal of copper and cadmium from aqueous solutions by activated carbon derived from Ceiba pentandra hulls. Journal of Hazardous Materials 129(1-3): 123-129. DOI: https://doi.org/10.1016/j.jhazmat.2005.08.018
Scholz, M. 2023. How activated carbon can help you-processes, properties and technological applications. Technologies 11(6): 153. DOI: https://doi.org/10.3390/technologies11060153
Siipola, V., Tamminen, T., Källi, A., Lahti, R., Romar, H., Rasa, K., Keskinen, R., Hyväluoma, J., Hannula, M. and Wikberg, H. 2018. Effects of biomass type, carbonization process, and activation method on the properties of bio-based activated carbons. BioResources 13(3): 5976-6002. DOI: https://doi.org/10.15376/biores.13.3.5976-6002
Tan, X., Liu, Y., Zeng, G., Wang, X., Hu, X., Gu, Y. and Yang, Z. 2015. Application of biochar for the removal of pollutants from aqueous solutions. Chemosphere 125: 70-85. DOI: https://doi.org/10.1016/j.chemosphere.2014.12.058
Yang, X., Wan, Y., Zheng, Y., He, F., Yu, Z., Huang, J., Wang, H., Ok, Y.S., Jiang, Y. and Gao, B. 2019. Surface functional groups of carbon-based adsorbents and their roles in the removal of heavy metals from aqueous solutions: A critical review. Chemical Engineering Journal 366, 608-621. DOI: https://doi.org/10.1016/j.cej.2019.02.119
Zhu, G., Deng, X., Hou, M., Sun, K., Zhang, Y. and Li, P. 2016. Comparative study on characterization and adsorption properties of activated carbons by phosphoric acid activation from corncob and its acid and alkaline hydrolysis residues. Fuel Processing Technology 144: 255-261. DOI: https://doi.org/10.1016/j.fuproc.2016.01.007