Effect of Calcination Temperature on Elemental Composition, Crystal Structure and Surface Morphology of Caulerpa lentillifera Residue
Main Article Content
Abstract
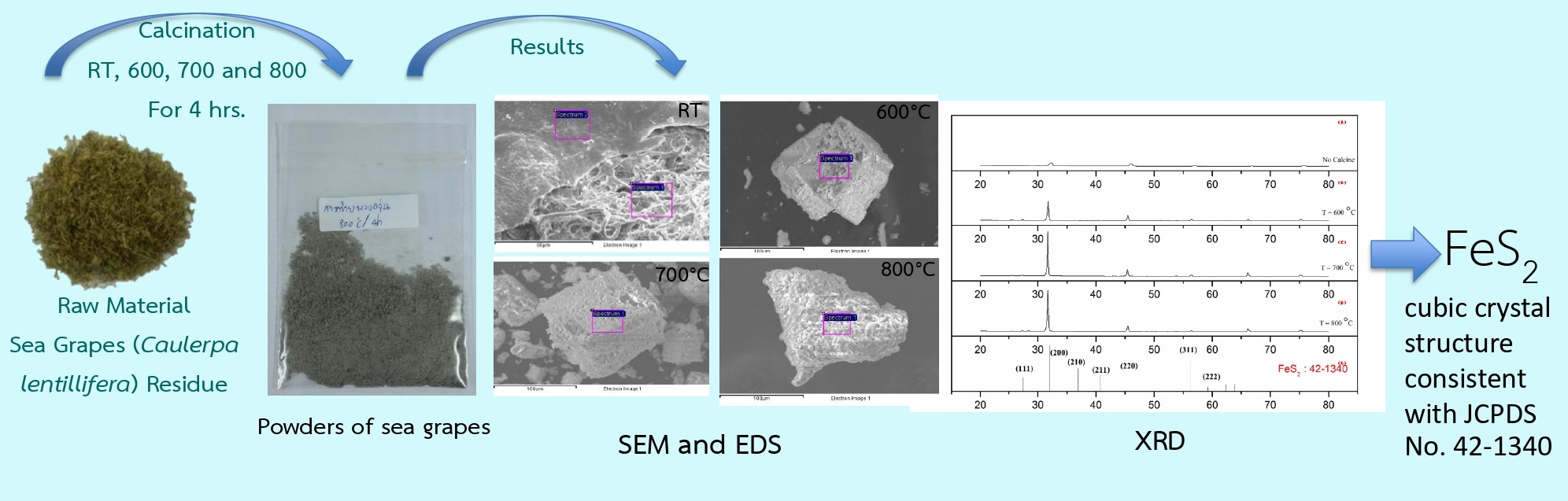
This research investigates the effects of calcination at room temperature and at 600ºC, 700ºC and 800 ºC for 4 hours on the elemental composition, crystal structure and surface morphology of Caulerpa lentillifera residue. The analytical techniques employed include energy-dispersive X-ray spectroscopy (EDS), X-ray diffraction (XRD) and scanning electron microscopy (SEM). Elemental analysis revealed the presence of 15 elements. The carbon and oxygen contents of the Caulerpa lentillifera residue decreased as the calcination temperature increased. Moreover, the concentrations of chlorine and sodium increased particularly at 800ºC, reaching 48.03 wt% and 29.25 wt%, respectively. XRD analysis at 600°C, 700°C and 800°C showed distinct diffraction peaks corresponding to the pyrite phase (FeS2) with a cubic crystal structure, specifically along the (111), (200), (220), and (311) planes, consistent with JCPDS No. 42-1340. SEM image indicated that increasing the calcination temperature resulted in a transformation from an amorphous fibrous morphology to well-ordered, fine crystalline structures. At 800°C, well-defined cubic geometric crystals were observed, consistent with the XRD results. These findings demonstrate that calcination temperature significantly influences both the elemental composition and crystalline structure of the biomass, indicating its potential for development as an adsorbent, catalyst, or ceramic material. The selection of an appropriate calcination temperature can determine the desired material properties, for instance, utilizing the increased sodium and chlorine content for adsorption applications, or employing the FeS2 phase as a catalyst.
Article Details
References
Mekasuk, C. and Thammaphiphol, S., 2017, Creating value added for Caulerpa lentillifera from upstream to downstream, J. Bus. Adm. Vis. (BAMJ). 9(2): 68–80. (in Thai)
Lertsongkram, A., Seaweeds Government Pharmaceutical Organization, Available Source: https://www2.gpo.or.th/Portals/6/Newsletter/RDINewsYr23No2-4.pdf, December 14, 2024. (in Thai)
Joshi, S., Kumari, R. and Upasani, V.N., 2018, Applications of algae in cosmetics: An overview, Int. J. Innov. Res. Sci. Eng. Technol, 7(2): 1269-1278.
Chantree, P., Martviset, P., Sornchuer, P., Thongsepee, N., Sangpairoj, K., Meemon, K., Niamnont, N., Tamtin, M. and Sobhon, P., 2023, Ethyl acetate extract of Halymenia durvillei induced apoptosis, autophagy, and cell cycle arrest in colorectal cancer cells, J. Food Sci. Nutr. 28(1): 69.
Wang, K., Peng, N., Lu, G. and Dang, Z., 2020, Effects of pyrolysis temperature and holding time on physicochemical properties of swine-manure-derived biochar, Waste Biomass Valorization. 11: 613-624.
Bahri, S., Upadhyay, R. and Harshwardhan, H., 2024, The effect of calcination temperature on the chemical and physical properties of corn straw ash, Int. J. Humanit. Soc. Sci. Manag. (IJHSSM). 4(4): 1062-1065.
Henríquez, R., Muñoz, E., Soto, G. and Mora, E., 2016, Single phase FeS₂ (pyrite) thin films prepared by combined electrodeposition and hydrothermal low temperature techniques, Int. J. Electrochem. Sci. 11: 4966–4978.
Qian, G., Xia, F., Joël, B., William M, S., Bei, J., Chen, G. and Allan, P., 2011, Replacement of pyrrhotite by pyrite and marcasite under hydrothermal conditions up to 220 °C: An experimental study of reaction textures and mechanisms, Am. Mineral. 96 (11-12): 1878-1893.
Kim, G., Ryu, J., Ryu, T., Kim, H.-I., Shin, J. and Cho, D.-W., 2024, Effects of calcination temperature on the adsorption ability of polyaluminum chloride (PAC) sludge-derived granules for As(V), J. Water Process Eng. 57: 104688.
Ferreira, H.M., Lopes, E.B., Malta, J.F., Ferreira, L.M., Casimiro, M.H., Santos, L.F., Pereira, M.F., Moço, D. and Gonçalves, A.P., 2021, Preparation and densification of bulk pyrite, FeS2., J. Phys. Chem. Solids. 159: 110296.